
Dry fog technology has become the gold standard for rapid, high-level disinfection in controlled environments.
When a laboratory undergoes scheduled maintenance, be it HVAC repairs, equipment installation, or structural touch-up,s the sterile integrity of the room is compromised.
This article explores the critical process of validating DryFog systems to ensure your facility returns to a Ready-to-Use state with zero microbial compromise.
In pharmaceutical, biotech, and high-security research labs (BSL-3/4), clean is not enough. After maintenance crews have entered a facility, the environment must be sterilized to a $10^{-6}$ reduction in microbial load.

DryFog systems, which utilize a fine mist of peracetic acid (PAA) and hydrogen peroxide ($H_2O_2$), are the preferred method. However, the system is only as good as its validation protocol.
Maintenance activities introduce specific risks that standard daily cleaning cannot address.

To meet regulatory standards (FDA/EMA), the DryFog process must follow a strict validation lifecycle.
Before running a cycle, you must verify that the DryFog hardware is installed correctly.

This includes checking air pressure lines, nozzle integrity, and ensuring the room’s HVAC dampers can be fully sealed to prevent sterilant leakage.
This phase tests the equipment’s ability to perform within defined limits.

This is the most critical step. It involves using Biological Indicators (BIs), typically Geobacillus stearothermophilus spores, to prove a 6-log reduction.
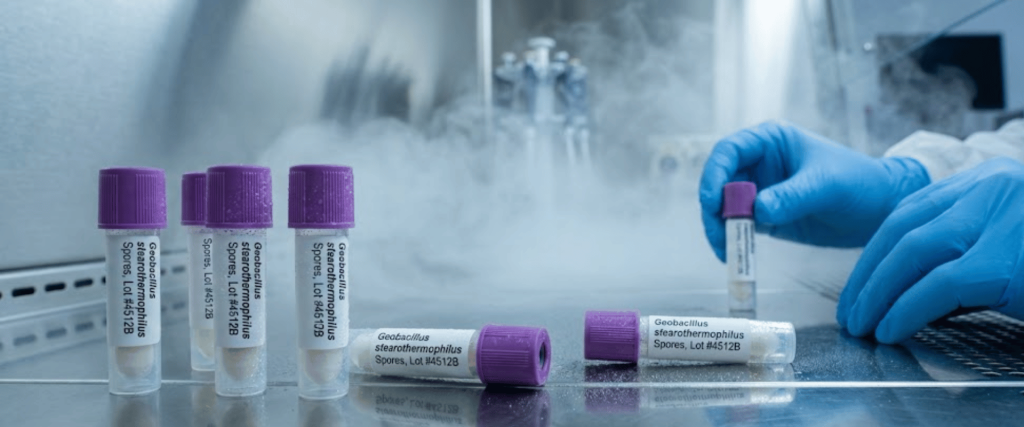
PQ must be performed in triplicate to ensure the results are repeatable and reliable.
A successful validation rests on four key variables, often referred to as the Cycle Development phase.
A primary concern during post-maintenance sterilization is the impact on newly installed equipment. DryFog is highly compatible with stainless steel, glass, and most plastics.

However, validation must include a review of sensitive sensors or soft materials introduced during maintenance to ensure no oxidative damage occurs.
This table positions DryFog as the superior choice for post-maintenance scenarios by comparing it against traditional methods.
| Feature | Dry Fog Technology | Vaporized H₂O₂ (VHP) | Manual Chemical Wipe | UV‑C Radiation |
|---|---|---|---|---|
| Disinfection Method | Ultra‑fine dry fog | Molecular vapor | Liquid chemical wiping | UV‑C light |
| Particle Size | 1–10 microns | Molecular vapor | N/A (liquid) | N/A (light) |
| Reach / Penetration | Excellent (shadow‑free) | Good | Poor (human error) | Poor (line of sight only) |
| Material Safety | High (non‑corrosive) | Moderate (oxidizing) | Low (residue risk) | High |
| Validation Ease | High (BI / CI compatible) | High | Low (subjective) | Moderate |
| Re‑entry Time | Rapid (30–90 minutes) | Moderate (2–4 hours) | Immediate | Immediate |
Robust validation of DryFog systems is the definitive bridge between facility maintenance and a return to sterile, compliant operations.
By integrating rigorous biological testing with real-time monitoring, labs can ensure a 6-log reduction in microbial load without compromising material integrity.
This systematic approach not only meets global regulatory standards but also safeguards the long-term reliability of your high-containment environment.
A standard sterilization cycle typically lasts between 60 and 90 minutes, though biological indicator (BI) incubation for final confirmation takes 24 to 48 hours.
Yes, because the dry particles are kept between 1–10 microns, they do not condense on surfaces, making the process non-corrosive for computers and sensors.
Geobacillus stearothermophilus spores are the industry standard for validating DryFog systems, as they are highly resistant to oxidative sterilants like peracetic acid.
Since 1992, Applied Physics Corporation has been a leading global provider of precision contamination control and metrology standards. We specialize in airflow visualization, particle size standards, and cleanroom decontamination solutions for critical environments.