
In pharmaceutical manufacturing, biotechnology, and semiconductor fabrication, the integrity of high-purity water (HPW) and water-for-injection (WFI) is paramount.
One of the most persistent threats to these systems is biofilm formation.
Unlike planktonic (free-floating) bacteria, biofilms are complex, sessile communities of microorganisms that adhere to surfaces, creating a self-protecting matrix that is notoriously difficult to eradicate.
It is a common misconception that ultrapure water systems, which are devoid of most nutrients, cannot support microbial life. In reality, many microorganisms are oligotrophs, capable of surviving and even thriving in low-nutrient environments.
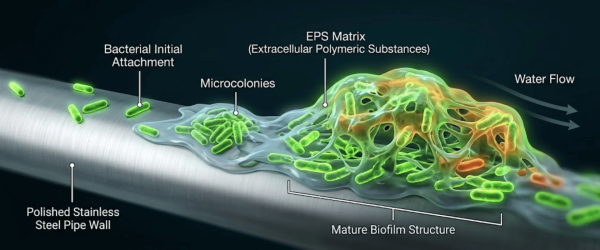
Once a single cell adheres to the internal surface of a pipe or storage tank, it begins to produce extracellular polymeric substances (EPS). This slime layer protects the colony from chemical sanitants and shear forces, leading to a resilient contamination source within the water loop.
The presence of biofilm in a pure water system is not merely a biological issue; it poses significant operational and regulatory risks.
Preventing biofilm requires a multi-faceted approach involving system design, material selection, and rigorous maintenance protocols.
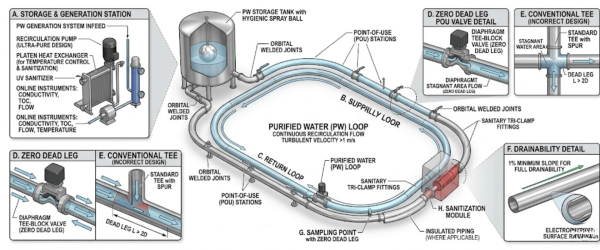
The physical layout of the water loop is the first line of defense.
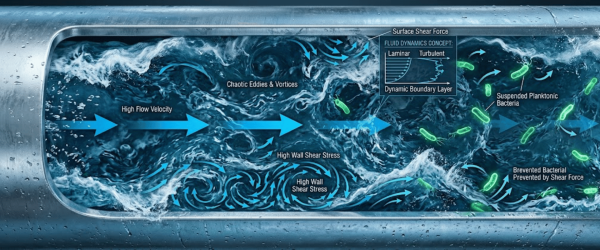
Maintaining a constant velocity is essential. Turbulent flow (typically defined by a Reynolds number > 4,000) creates shear forces that discourage bacterial attachment.
Systems should be designed to run continuously rather than using start-stop cycles, which allow for stagnation.
Regular sanitization cycles are necessary to prevent the transition from initial attachment to a mature biofilm matrix.

| Method | Primary Mechanism | Best Application | Key Advantage |
|---|---|---|---|
| Thermal Sanitization | High heat (> 80 °C) denatures microbial proteins | WFI (Water for Injection) loops | No chemical residues; highly reliable |
| Ozonation | Strong oxidation breaks down cell walls and EPS | Storage tanks and distribution loops | Very powerful oxidant; reduces TOC |
| UV Radiation | DNA/RNA disruption prevents microbial replication | Point of use and ozone destruction | Continuous operation; no taste or odor impact |
| Hydraulic Shear | High‑velocity turbulent flow (Re > 4000) | Continuous loop distribution | Prevents initial bacterial attachment |
| Chemical Cleaning | Acid/alkali washing removes organic and inorganic scale | Periodic deep system cleaning | Effective against mature, stubborn biofilms |
Verification is the final component of a robust contamination control strategy.

Traditional plate counts are often insufficient because they only detect planktonic bacteria that have already broken off from a biofilm.
Preventing biofilm in pure water systems is an ongoing process of vigilance.
By combining hygienic by design engineering with rigorous sanitization and advanced monitoring, facilities can ensure the consistent delivery of high-purity water that meets the stringent requirements of modern industry.
Addressing biofilm risks is not just a maintenance task; it is a fundamental requirement for process stability and consumer safety.
Biofilms are formed by oligotrophic bacteria that are specifically adapted to survive in low-nutrient environments. Even a single microbe can adhere to a pipe surface and secrete a protective slime layer (EPS), allowing it to thrive and multiply despite the lack of traditional nutrients.
The 316L Rule (or the 2-diameter rule) is a design standard used to eliminate dead legs in piping. It states that any branch or T-junction in a water loop should have a length no greater than two times its pipe diameter to ensure constant water movement and prevent stagnation.
No. While UV radiation is excellent at disrupting the DNA of free-floating (planktonic) bacteria to prevent replication, it cannot penetrate the thick, protective matrix of an established biofilm attached to a surface. UV is a preventative tool, not a removal method for mature contamination.
Since 1992, Applied Physics Corporation has been a leading global provider of precision contamination control and metrology standards. We specialize in airflow visualization, particle size standards, and cleanroom decontamination solutions for critical environments.