In industries like semiconductor manufacturing and biopharmaceuticals, clean is a relative term.
While visible particles were once the primary concern, the focus has shifted to Airborne Molecular Contamination (AMC) and liquid-phase molecular impurities.
In high-purity chemical storage, even parts-per-billion (ppb) or parts-per-trillion (ppt) levels of unwanted molecules can lead to catastrophic yield loss, catalyst poisoning, or compromised drug efficacy.
Managing molecular contamination is not just about filtration; it is a holistic discipline involving materials science, atmospheric control, and rigorous monitoring protocols.
Molecular contaminants are non-particulate chemical species that can be categorized into four primary groups based on their behavior and impact.

Contamination in storage typically arises from three areas.
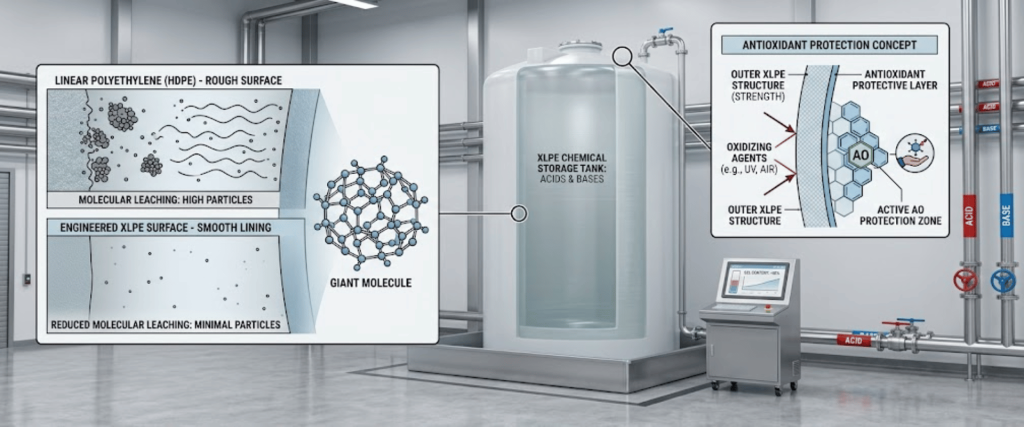
Selecting the right Material of Construction (MoC) is the first line of defense. Standard industrial plastics often contain plasticizers and stabilizers that leach into high-purity chemicals.
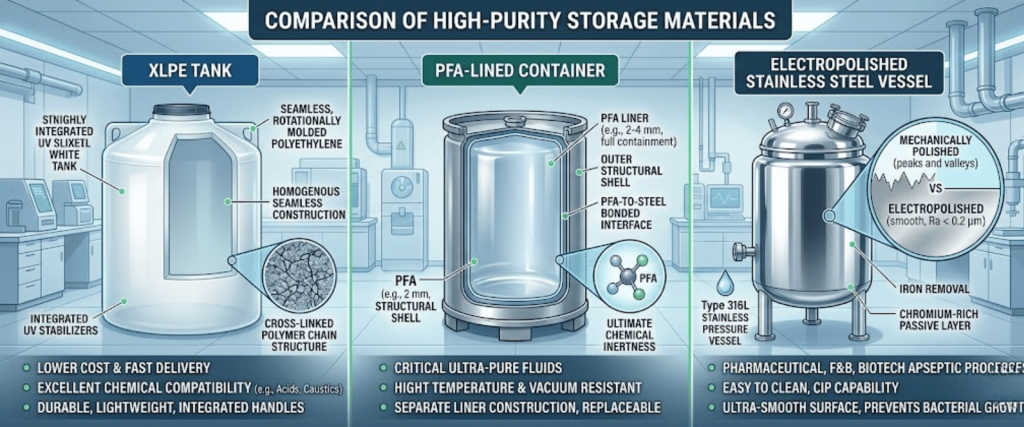
Common for bulk storage of acids and bases. XLPE offers superior structural integrity and is often treated with antioxidant resins to reduce molecular leaching by up to 75%.
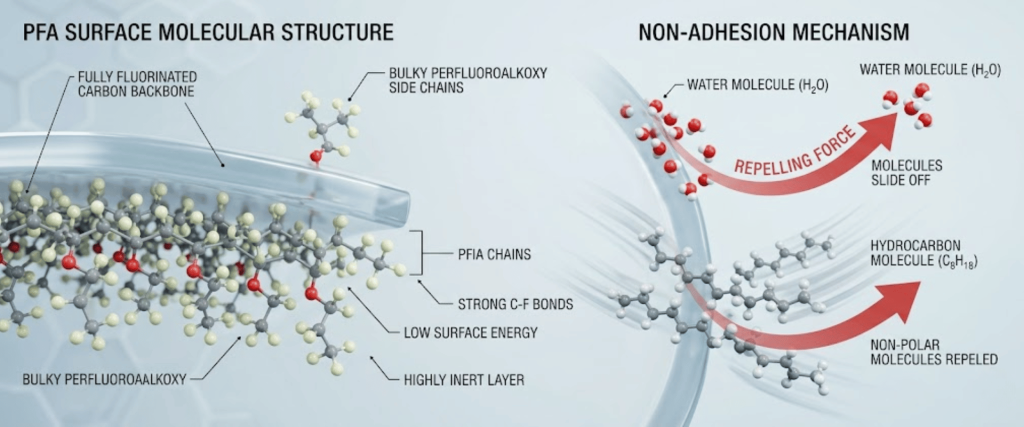
The gold standard for ultra-high purity (UHP) chemicals. PFA is chemically inert and has an extremely smooth surface that prevents molecular adhesion and biofilm growth.
Note: SEMI C90 provides the standard test methods for PFA materials used in liquid chemical distribution.
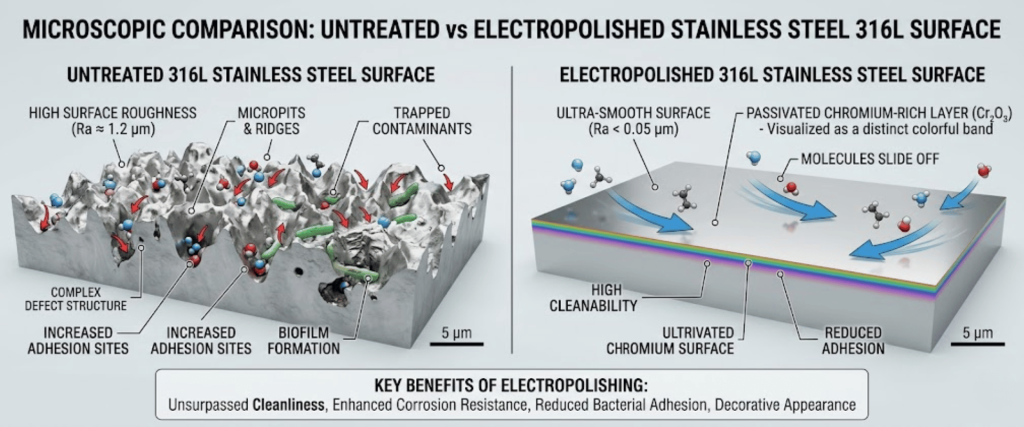
Used for solvent storage. The electropolishing process removes surface irregularities, reducing the surface area where molecules can trap and later outgas.
To maintain the integrity of chemicals during storage, facilities must implement active engineering controls.
By maintaining a positive pressure of ultra-high purity nitrogen in the headspace of a storage tank, you prevent atmospheric moisture and oxygen from contacting the chemical.

This is critical for preventing the formation of $H_2O$ and $CO_2$ related contaminants.
Standard HEPA filters capture particles but are useless against gas-phase molecules. Advanced storage systems are utilized.
Following international standards ensures that your storage protocols meet global quality benchmarks.
| Standard | Scope | Key Focus |
|---|---|---|
| SEMI F31 | Bulk Chemical Distribution | Design and operation of bulk systems to minimize contamination |
| SEMI F57 | Polymer Materials | Specifications for polymers used in UHP liquid distribution |
| ISO 14644-8 | AMC Classification | Classification of airborne molecular contamination levels |
| SEMI E49 | UHP Piping | Performance guides for piping systems and subassemblies |
Managing molecular contamination in high-purity chemical storage requires a shift from reactive cleaning to proactive prevention.
By combining inert materials like PFA, rigorous SEMI compliance, and real-time CRDS monitoring, manufacturers can safeguard their yields and ensure the highest product quality.
Molecular contamination typically comes from three main sources: outgassing of storage materials, permeation of atmospheric gases through tank walls, and cross-contamination from residual chemicals in distribution lines.
PFA (Perfluoroalkoxy) is the industry gold standard because it is chemically inert and has an extremely smooth surface. This prevents molecules from sticking to the walls and ensures no harmful substances leach into the chemicals.
Nitrogen blanketing maintains positive pressure in the tank’s headspace using ultra-pure nitrogen. This creates a physical barrier that stops moisture, oxygen, and other airborne molecules from entering and reacting with the stored chemical.
The most critical benchmarks are the SEMI standards (such as SEMI F57 for polymers and SEMI C90 for testing) and ISO 14644-8, which classifies airborne molecular contamination levels in cleanroom environments.
Since 1992, Applied Physics Corporation has been a leading global provider of precision contamination control and metrology standards. We specialize in airflow visualization, particle size standards, and cleanroom decontamination solutions for critical environments.