
In the landscape of modern pharmacology and healthcare, the handling of hazardous drugs (HDs) presents a dual challenge: maintaining the sterility of the medication while ensuring the absolute safety of the healthcare professional.
USP General Chapter <800> was developed to address these risks, providing a rigorous framework for the Handling of Hazardous Drugs in Healthcare Settings.
A cornerstone of this compliance is the validation of airflow patterns within sterile compounding areas.
Precision airflow visualization, often referred to as smoke studies, is not merely a recommendation; it is a technical necessity to prove that engineering controls are performing as intended.
USP <800> applies to all healthcare personnel who help handle HD preparations and all entities that store, prepare, transport, or administer HDs. Unlike previous standards that focused primarily on the patient, USP <800> places a heavy emphasis on occupational safety.
Hazardous drugs ranging from antineoplastic agents to certain hormones can cause reproductive toxicity, organ damage, or even cancer in workers if exposure occurs through inhalation or skin contact.

To mitigate these risks, the standard mandates the use of Containment Primary Engineering Controls (C-PECs), such as Biological Safety Cabinets (BSCs) and Compounding Aseptic Containment Isolators (CACIs).
A C-PEC is designed to provide an ISO Class 5 environment. It relies on a unidirectional (laminar) flow of HEPA-filtered air to sweep contaminants away from the compounding area.
The most critical concept here is First Air, the air exiting the HEPA filter that has not yet been interrupted by any items (vials, needles, or hands) in the workspace.
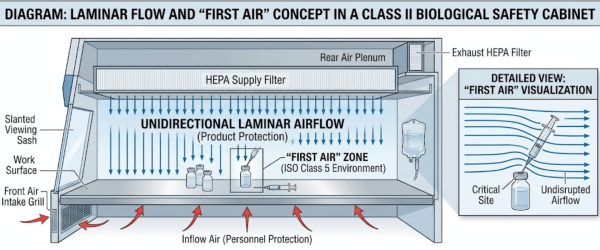
If the airflow is turbulent or blocked, dead zones can form, allowing hazardous vapors or particles to linger or escape into the worker’s breathing zone.
While particle counters and velocity meters provide quantitative data, they cannot show the path of the air. This is where airflow visualization becomes indispensable.
A smoke study provides visual evidence that the C-PEC is maintaining a proper air curtain and that the inward inflow velocity is sufficient to prevent HD particles from escaping the cabinet.

Placement of equipment within a BSC can inadvertently cause turbulence.

Airflow visualization allows pharmacists to see exactly how air moves around objects, enabling them to optimize the layout of their workspace to maintain a sterile sweep.
USP <800> requires that personnel be trained in proper HD handling.

Watching a smoke study helps staff understand how their hand movements affect airflow, teaching them to work cleanly without breaking the laminar flow.
Not all smoke is created equal. For USP <800> and USP <797> compliance, the method used to visualize air must not introduce its own contamination.
Using smoke sticks or chemical smoke is increasingly discouraged in sterile environments due to the potential for particulate residue and toxicity.
To remain compliant with USP standards, airflow visualization should be performed:
Navigating USP <800> compliance is a continuous process of validation.
By prioritizing precision airflow visualization, healthcare facilities do more than just check a regulatory box; they build a transparent culture of safety.
The use of advanced metrology tools, like ultrapure cleanroom foggers, ensures that the invisible barrier between hazardous drugs and the people who handle them remains impenetrable.
Airflow visualization, also known as a smoke study, provides visual proof that your containment systems are effectively protecting workers from hazardous drug particles and keeping the workspace sterile.
To stay compliant, you should perform airflow visualization at initial installation, every six months during routine recertification, or whenever your equipment is moved or repaired.
High-purity foggers (like Ultrasonic or LN2 models) are the gold standard. Unlike chemical smoke sticks, they are completely residue-free and won’t contaminate your sterile compounding environment.
Since 1992, Applied Physics Corporation has been a leading global provider of precision contamination control and metrology standards. We specialize in airflow visualization, particle size standards, and cleanroom decontamination solutions for critical environments.